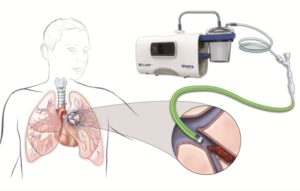
Insera Therapeutics announced recently that the first patient with acute pulmonary embolism has been treated with its flagship cyclical aspiration system, CLEAR. Earlier last month, the first two stroke patients were treated using the same platform. The company received CE mark approval for its flagship product, the CLEAR aspiration system, in March 2019.
“Due to the large clot burden in the lung arteries, the 65-year old gentleman presented with right-sided heart failure and was in critical condition. Traditional treatments using blood thinning medications such as anti-coagulation and thrombolytic medications were contraindicated in this patient. We successfully treated this patient with massive bilateral acute pulmonary embolism using Insera’s CLEAR aspiration system,” said Luka Novosel, interventional radiologist at the Sisters of Charity Hospital, Zagreb, Croatia. “I am very pleased with the ease of use of cyclical aspiration and its ability to rapidly ingest large amounts of clot. It also provides a safer mechanical means of removing life-threatening blood clots without the bleeding risk associated with blood thinning medications.”
Insera’s first-in-human success in treating pulmonary embolism comes on the heels of the recent announcement of the EXTRACT-PE trial results presented at the Vascular InterVentional Advances (VIVA) meeting (4–7 November, Las Vegas, USA). This trial advances treatment options for the industry as it establishes the use of vacuum aspiration in patients with acute pulmonary embolism.
The CLEAR aspiration system has European regulatory approval for the aspiration of blood clots, and is indicated for use for acute ischaemic stroke secondary to large vessel occlusive disease, as well as the aspiration of blood clots from arteries (e.g. peripheral artery disease, pulmonary embolism, and coronary artery disease), and for the aspiration of blood clots from veins (e.g. in patients with deep venous thrombosis, cerebral venous sinus thrombosis or patients with renal failure who have clotted haemodialysis grafts).
“To date, we have successfully used the CLEAR aspiration system in nine stroke patients. I am very pleased with the higher trend of first-pass recanalisation using cyclical aspiration. For example, in our very first patient, we had a 6 minute procedure time from groin to recanalisation with TICI 3 on first-pass (complete vessel re-opening on the first operative attempt),” said Vladimir Kalousek, accredited interventional neuroradiologist at the Sisters of Charity Hospital, Zagreb, Croatia who has pioneered the use of cyclical aspiration.
“There is a huge unmet need for innovative devices for the removal of life-threatening blood clots. Our unique and proprietary cyclical aspiration technology has been shown to improve first-pass recanalisation, reduce distal embolisation, and has an improved safety profile at the time of the procedure,” said Vikram Janardhan, chief executive officer of Insera Therapeutics. “In the USA alone, it is estimated that every year there are around 225K patients with ischaemic strokes due to large vessel occlusions (LVOs), 300K patients with PE, 600K patients with DVTs, and 100K patients with acute limb ischaemia.”












