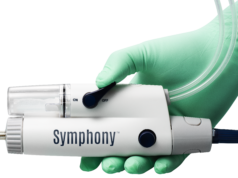
Fist Assist Devices has announced a two-year affiliation with Airos Medical to commercialise and launch sales of the Fist Assist FA-1 device in the USA. The Fist Assist FA-1 is a wearable, patent-protected, intermittent compression device that recently received US Food and Drug Administration (FDA) clearance for arm vein massage and vein circulation.
Fist Assist Devices has announced a two-year affiliation with Airos Medical to commercialise and launch sales of the Fist Assist FA-1 device in the USA. The Fist Assist FA-1 is a wearable, patent-protected, intermittent compression device that recently received US Food and Drug Administration (FDA) clearance for arm vein massage and vein circulation.
“We are thrilled to combine forces with Airos Medical for the official launch of the Fist Assist FA-1 device in the USA. This is a huge milestone as we now have a committed, dedicated, and exceptional commercialisation arm that is poised to deeply penetrate the US market,” said Tej Singh, chief executive officer and founder of Fist Assist Devices. “As a vascular surgeon, I have worked with pneumatic devices for lymphoedema distributed by Airos for many years, and I am pleased to see Fist Assist join the Airos product line of innovative pneumatic products. I am very excited to finalise this alliance and look forward to working with Airos Medical to grow the US market for the FA-1 device. Our goal at Fist Assist is to temporarily increase arm vein circulation and relieve minor muscle aches and soreness while making this innovative product available to as many users as possible. This alliance will help us achieve that!”
“With the recent clearance for arm vein massage and vein circulation, we are excited to distribute and commercialise the innovative FA-1 device,” said Chris Allerton, vice president of business development of Airos Medical. “We have been watching the Fist Assist technology for the past three years and patiently awaiting market clearance as I believe this is a groundbreaking, market-changing, innovative, person-focused product that will improve user quality of life throughout the USA. Plus, as a pneumatic compression device the FA-1 is a natural extension of our current product line and point of sale interaction. We are thrilled to be selected to launch and commercialise this technology!”