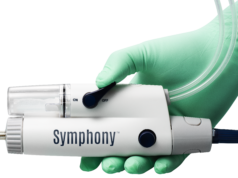
InterVene recently announced that it has received Breakthrough Device designation from the US Food and Drug Administration (FDA) for the company’s BlueLeaf endovenous valve formation system.
According to the company, BlueLeaf is the first catheter-based solution developed for deep vein reflux which does not require an implant. Clinical research is underway in three separate study programmes worldwide, including an early feasibility study in the USA.
The BlueLeaf system is designed to correct one of the underlying causes of chronic venous insufficiency (CVI), deep vein reflux, by forming new, autologous vein valves out of the patient’s vein wall tissue via an endovascular, ultrasound image-guided approach. To date, over 25 patients with varied anatomy, disease severity, and aetiology have had new vein valves formed by US and international investigators using the BlueLeaf system, InterVene reports. They add that both monocuspids and bicuspid valves have been formed.
“This is a meaningful milestone for the company which offers the potential for a more timely clinical research and regulatory process for BlueLeaf. Given the debilitating nature of DVR and the lack of an effective treatment option for these patients, we are pleased with the FDA’s decision and look forward to productive collaboration with the agency. In addition, potential economic and reimbursement benefits remain an ongoing process which we will follow closely as we plan and develop the next phase of our clinical research,” noted InterVene CEO Jeff Elkins.
“The FDA’s designation of the BlueLeaf endovenous valve formation system as Breakthrough is an important recognition of both the tremendous unmet need for deep vein reflux patients and the potential of the novel non-implant valve technology as treatment for the severe symptoms they frequently experience. Solutions to deep vein reflux have been elusive despite decades of research, and InterVene’s autologous, endovenous approach is worthy of prioritisation, so the designation is a fitting and positive development,” said William Marston, the George Johnson Jr distinguished professor of Vascular Surgery at the University of North Carolina School of Medicine (Chapel Hill, USA).