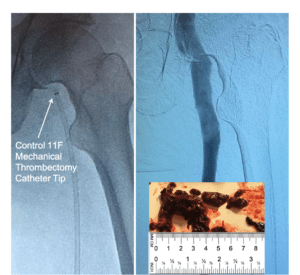
 Control Medical Technology recently announced that their US Food and Drug Administration (FDA)-cleared Control 11F mechanical thrombectomy system has been used to remove large blood clots from patients with deep vein thrombosis (DVT).
Control Medical Technology recently announced that their US Food and Drug Administration (FDA)-cleared Control 11F mechanical thrombectomy system has been used to remove large blood clots from patients with deep vein thrombosis (DVT).
“The Control 11 French mechanical thrombectomy system can remove large clots,” said Shawn Fojtik, president of Control Medical Technology. “Patients, clinicians, and payors need more tools to remove diverse thrombus without excessive expense and complex capital equipment. A disposable US$2,000–15,000+ device per patient technology is not the only way to remove blood clots, especially if it sucks too much blood.”
During thrombectomy and embolectomy procedures, clinicians typically track a catheter to the thrombus/emboli and then use a basic syringe or high-performance aspirator/electric pump to pull the clot/emboli out. Basic syringes lack speed, force, volume, and control. Electric pumps improve force but are complex and reported to suck too much blood with 7+ French (~2.25mm+) outer diameter catheters.
“Reducing blood loss is important when removing large clots and emboli,” continued Fojtik. “Large catheters can remove blood too fast when using electric pumps. Our new FDA-cleared system is designed to improved control with a wide range of catheters. We look forward to seeing improved clot targeting, capture, maceration, and removal improve patient care.”












