This advertorial is sponsored by Inari Medical
“We need to eliminate symptoms as fast as possible—it is not OK just to make things a little better,” says Rick de Graaf (Clinical Centre of Friedrichshafen, Friedrichshafen, Germany), setting out why intervention in acute deep vein thrombosis (DVT) is important. De Graaf cites quality of life and economic burden as key factors. “That does not allow us to be too risky,” he continues, commenting that avoiding complications, such as bleeding or recurrent DVT are fundamental to any intervention.
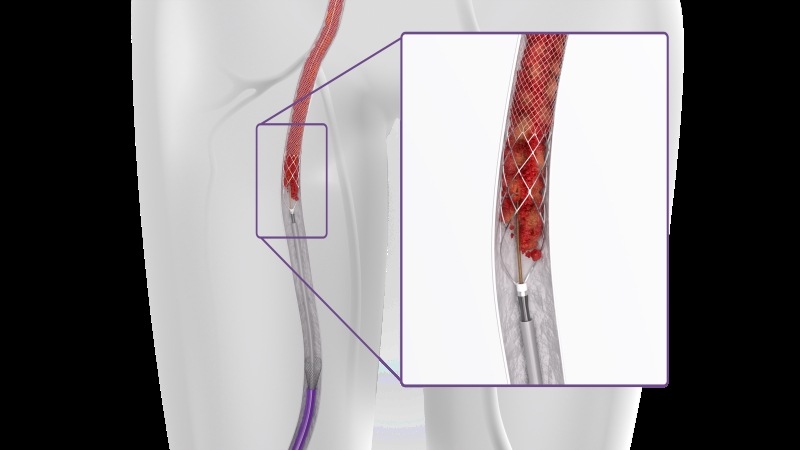
According to De Graaf it does not make sense to use a device that takes out, 50, 60 or 70% of thrombus. He argues that full removal should be the ultimate strategy to optimise clinical outcome.
De Graaf cites ClotTriever (Inari Medical) as one such device that makes fast, safe and efficient thrombectomy possible. Outcomes of the device in a real-world DVT population have been demonstrated through the ongoing CLOUT registry, a 500-patient, prospective, multicentre, all-comer study, taking place at 43 centres across the USA, making it the largest mechanical thrombectomy study in the field of DVT. Results have so far been shown out to 30 days, with follow-up planned out to two years. The trial’s primary endpoint is technical success, defined as complete or near-complete (>75%) removal of thrombus by core lab-assessed Marder score.
“That is spectacularly high when you compare that to other devices and benchmarks,” says David Dexter (Sentara Vascular Specialists, Norfolk, USA) an investigator in the study, of the primary endpoint goal. However, he offers the view that investigators saw ClotTriever as likely to hit this target, “because of its purely mechanical nature in the acute phase and maybe in the subacute arm”.
Results out to 30 days, presented at The VEINS 2022 (30–31 October, Las Vegas, USA), demonstrate that the primary endpoint (>75% thrombus removal), was achieved in 91.2% of patients, while nearly two thirds of patients had 100% of clot removed. The intermediate number of 90% clot removal was seen in around 70% of patients.
Safety was also assessed, and the study has shown a serious adverse event (SAE) rate of 0.2%. Added to this, most patients were treated in a single session (99.4%), with minimal blood loss (40ml) and minimal use of thrombolytic therapy (0.4%). From an improvement standpoint, Dexter says, the first goal is restoration of flow. At baseline only 27.6% of patients in CLOUT had flow, but 30 days post-treatment, 85% of patients had some or all flow restored.
For Steven Abramowitz (Medstar Health, Washington DC, USA), putting these data in context with previously published studies is an important next step to guide future treatment decisions, in line with the universal goal to “function in an evidence-based world”. To achieve this aim, data from CLOUT have been compared to those from the ATTRACT trial, which assessed the use of pharmacomechanical catheter-directed thrombolysis, in a propensity-matched study. “ATTRACT is one of the fundamental reference trials in the USA, and I think globally, for guiding intervention for DVT,” says Abramowitz. The analysis looked at 153 pairs of similarly matched patients within the two trials based on 11 baseline covariates.
Results show better thrombus extraction with the ClotTriever system, with nearly 88% of the CLOUT patients achieving thrombus clearance ≥75%, compared to 66% who received an intervention in ATTRACT. There was also a significant difference in the number of patients with 100% thrombus clearance, totalling 58% in CLOUT, compared to 30% in ATTRACT. The analysis also compared reduction in Villalta Score—an indicator of the severity of post-thrombotic syndrome in lower-extremity DVT—recorded in the two trials, showing a significant decrease at 30 days in CLOUT with 27% of patients with a Villalta Score ≥5 vs. 40% in ATTRACT.
“Propensity matched analysis shows us that contemporary interventional strategies with ClotTriever do stand their ground and have comparable, if not potentially better results than those patients treated with interventional options available at the time of ATTRACT enrolment,” says Abramowitz.












