Tag: venous stent
US FDA approves first deep venous stent indicated for IVC and...
Gore today announced US Food and Drug Administration (FDA) approval of the Gore Viabahn Fortegra venous stent, which the company notes is the first...
Gore announces US FDA approval for Fortegra venous stent
Gore has announced the US Food and Drug Administration (FDA) approval of the Gore Viabahn Fortegra venous stent—previously known as the Gore Viafort vascular...
Prognostic tool for CVO stenting set to enhance therapeutic decision-making and...
With the aims of predicting and comparing venous stent outcomes, aiding in communication with patients, and enhancing therapeutic decision-making, researchers have proposed an anatomical...
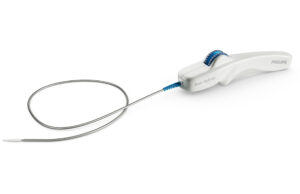
Philips launches Duo venous stent system for treatment of symptomatic venous...
Royal Philips today announced the first implant of the Duo venous stent system, an implantable medical device indicated to treat symptomatic venous outflow obstruction...
IVC 2024: Choosing the right stent for the right situation
Patrick Muck (Cinncinatti, Ohio) provided a state of the venous stent landscape during the 2024 International Vein Congress (IVC; April 18–20) in Miami.
The TriHealth...
Venous stents remain durable following pregnancy
Iliac vein stents tolerate a gravid uterus well and a possible future pregnancy should not contraindicate their usage in the treatment of pelvic venous...
Bentley enters venous world with launch and first-in-man of BeYond stent
Bentley has announced receipt of the CE mark for its BeYond venous self-expanding stent system. The company will initially launch the stent to an...
Vesper Medical completes US$10.5m Series A financing
Vesper Medical, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, has announced that it completed its Series A financing, totaling...
UIP audience votes against stenting as standard for deep vein obstruction
The deep venous world is wrestling with the question: To stent or not to stent. While the availability of new specialised venous stents is...
blueflow Venous Stent receives CE mark approval
The blueflow Venous Stent (plus medica) received the CE mark in January 2018.
The news was announced via a company release in March.
The blueflow Venous...
Veniti announces Boston Scientific distribution agreement for Vici venous stent
Boston Scientific will distribute Veniti’s Vici venous stent under a limited global distribution agreement. The terms of the agreement and specific regions and countries...
Final VIVO-EU results show nearly 90% freedom from occlusion rate for...
Gerard O’Sullivan, Department of Radiology, University Hospital Galway, Ireland, presented the results of the VIVO-EU prospective study of the Zilver Vena venous stent (Cook...