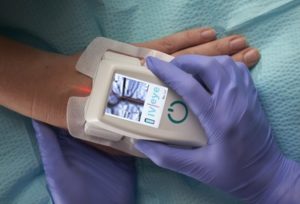
CorVascular has entered into a distribution agreement with Novarix. The IV-eye, developed and manufactured in the UK by Novarix, is a near infrared (NIR) vein imaging device designed to aid healthcare professionals in finding suitable peripheral veins for both cannulation and venipuncture.
Under the agreement between the companies, CorVascular will exclusively market the IV-eye to national and large regional distributors. A key component of the arrangement makes CorVascular responsible for introducing the IV-eye to resellers throughout all healthcare segments of the US market.
“We have been looking at vascular visualisation technologies with the goal to broaden our product line, and the IV-eye is the perfect solution,” said Spencer Lien, chief executive officer of CorVascular. “We now have the ideal product for distribution into several market segments: long-term care, surgery centres, hospitals, and teaching institutions. All of which can benefit from a low cost, simple to use vein finder to help any staff member quickly obtain access.”
Patients with difficult intravenous access are frequently subjected to repeated attempts by multiple practitioners due to failed intravenous attempts and are more likely to experience treatment delays. The average time requirement for peripheral intravenous cannulation has been reported at 2.5 to 16 minutes, with difficult access requiring as much as 30 minutes. A delay in establishing vascular access can result in a delay in the administration of fluids and/or medications as well as delays in diagnosis and initiation of treatment. Multiple attempts at attaining vascular access result in frustration and a loss of productivity by the treating team as well as a poor patient experience.
John Scott, president and chief executive officer of Novarix, says, “We are proud to be partnered with CorVascular. Their proven ability to successfully introduce new products into the US market was a key factor in choosing them. They have the right distribution experience to quickly get the IV-eye into the hands of American distributors, while we continue to build momentum for the product around the world.”