
Continuing work to develop an improved percutaneous valvuloplasty procedure “is promising”, said Johann Chris Ragg (Angioclinic Vein Centers, Berlin, Munich, Germany, and Zurich, Switzerland), as he updated delegates on the technique at the annual American College of Phlebology meeting (ACP; 2–5 November, Austin, USA). He also argued that now is the time to “prepare the stage” for this treatment, primarily by recognising early phase reflux as its own specific disease.
Valve zone dilation is one of the three ways by which primary venous insufficiency can develop (alongside congenital valve defects and inflammatory valve degeneration). This leads to consecutive local overload and progression of venous incompetence. “If dilation is the problem, compression could be the solution,” Ragg told ACP delegates, by treating the temporary reflux that marks the beginning of the disease process. In the case of percutaneous valvuloplasty, this can be achieved using perivenous hyaluronan gel to shape the vein and restore flow.
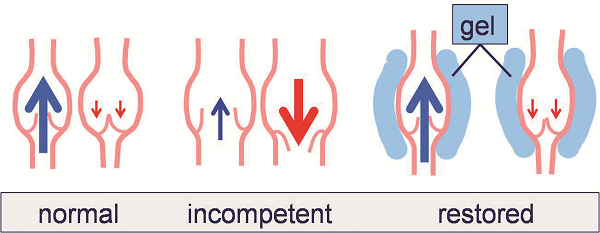
A “good” percutaneous valvuloplasty procedure requires the selection of patients with failing, but still intact and mobile, cusps, Ragg explained. The hyaluronan gel must have an appropriate balance of injectability (which improves as viscosity is reduced) and durability (which improves as viscosity increases). Finally, the injection technique should place minimal amounts of gel as close as possible to the vein wall, in order to make the procedure more effective and more affordable.
The first two trials of percutaneous valvuloplasty conducted were very similar in design, but used two different gels. The first study took place in 2013 and used Macrolane (VRF 30), which is a low-cost, large-particle (>1mm) gel previously approved for breast enhancement procedures. The second trial ran from 2014 to 2015 and used Princess (Croma/Austria), which is a smooth, small-particle (<0.2mm) gel approved for aesthetic use.
The first study included 23 patients (15 female and eight males aged 38–67 years). The second study enrolled 18 patients (12 females and six males aged 34–69 years). Both trials were for proximal valve incompetence of the greater saphenous vein. The patient cohort for the first trial had vein diameters of 7–11.5mm with a mean diameter of 8.6mm. The cohort for the second trial had vein diameters of 7–11.8mm with a mean diameter of 8.9mm. Follow-up for both trials went out to 52 weeks and used ultrasound 3D scans.
Initial haemodynamic success was achieved in 92.6% (n=22) of Macrolane patients and 100% (n=18) Princess patients. A second session was required for 31.8% (n=7) of Macrolane patients and 27.8% (n=5) of Princess patients. Supplementary injections at one year were given to 31.8% (n=7) of Macrolane patients and 22.2% (n=4) of Princess patients. At one year follow-up, 100% of both groups had orthograde flow. The volume of gel used was higher in the Macrolane group (12–35ml, mean 19.4ml) than in the Princess group (4–9ml, mean 6.9ml). As for cost, Macrolane was much cheaper (US$18/ml) than Princess (US$120/ml), with a mean procedure cost of US$350 versus US$830.
A third study, conducted in 2017, evaluated hyaluronan gel, which Ragg described as the next step in the “evolution of injectable gels”. The average human has approximately 15g of hyaluronan in their body, one third of which is degraded and synthesised every day. Linkage of monophasic hyaluronan molecules with sorbitol is one way in which the viscosity, stability and persistence of injectable products can be modified.
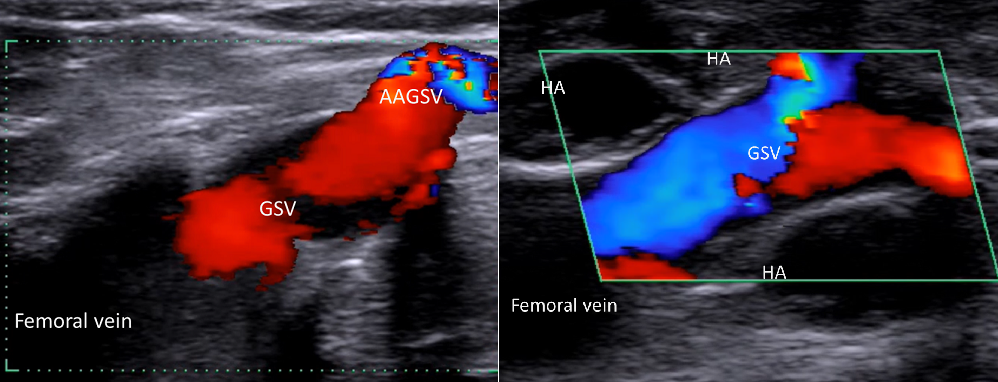
Ragg explained that the 2017 trial used this approach to inject hyaluronan gel with access site local anaesthesia and ultrasound monitoring, with no bandages or compression stockings used afterwards. Twelve patients (six female and six male aged 32–54 years) with proximal valve incompetence of the greater saphenous vein were treated. Vein diameters ranged from 7–12mm with a mean of 8.3mm. Orthograde flow was established in 100% (n=12) of cases, with no migration observed at three months. The gel volume used was 3–6.2ml with a mean of 4.7ml, and the potential mean cost was US$260—less gel and lower costs than in both the Macrolane and Princess trials.
“If you can modify a vein’s diameter by using a natural substance with a simple small-needle injection, big question marks arise concerning many kinds of vein surgeries,” Ragg suggested. “For example, thermal occlusion, sclerotherapy and gluing are all just killing veins with varying levels of risk.”
“Percutaneous valvuloplasty is a simple procedure for anyone who is used to ultrasound-guided punctures,” Ragg continued. “It is minimally invasive, easily adjustable and rechargeable, and is suitable for the early stages of vein valve insufficiency. Work to simplify injections and use smaller volumes is promising, and we now need to prepare the stage by recognising early phase reflux as a specific disease.”













Dear Vascular News Team!
It seems to be a very interesting an innovative way to treat certain forms of GSV incompetence.
I would like to know if there is some experience with treating the SSV by this way?
Could you please inform me what is the brand name of the hyaluronic gel used n the third trial and where it is possible to get it?
Kind regards,
Dr. Andreas Franczak
Dear Dr Ragg
Who writes to him is Dr. Enrique Ferracani of Argentina, creator of the patent of Idea Internal Laser Valvuloplasty (USA). Your contribution is useful but allow me a comment to enrich your presentation. Only the Valvuloplastias are effective, both internal SIL and external Ragg method in early stages, as in Class 3 -4 APEC the valve no longer exists or is so destroyed that is imposinble its Arrangement. The method Kevlar, of my authorship describes as in fact both you and me, what we do is to generate a resistance to the front of reflux pressure which consumes the reflux during the maneuver of Valsalva cordially
E Ferracani MD
corrections :
ILV Internal LASER valvuloplasty
EVLAR Endovascular LASER Remodeling
US Patent number 89851212
DATE march .24.2015
Estimado Dr. Ragg:
parecería algo prometedor por lo simple y económico, como cirujano plástico de Argentina que realizo bioimplantes naturales como sintéticos (grasa autóloga, metacrill , ácido hialurónico etc) me resultaría facíl, estoy ensayando otros productos más duraderos en el tiempo.
Saludos cordiales.
Dr Marcelo Almada
http://www.cirujanodralmada.com.ar