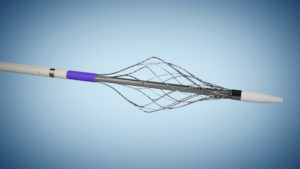
Surmodics recently announced that it has acquired privately-held Vetex Medical Limited. The Galway, Ireland-based medical device developer and manufacturer has focused exclusively on venous clot removal solutions. The transaction expands Surmodics’ thrombectomy portfolio with a second US Food and Drug Administration (FDA) 510(k)-cleared device, the ReVene thrombectomy catheter.
The ReVene mechanical thrombectomy catheter is specifically designed to remove large, mixed-morphology blood clots commonly found with venous thromboembolism (VTE). According to a press release, the device’s dual action technology efficiently removes mixed-morphology clot in a single session, minimising the need for thrombolytics and without capital equipment.
“This acquisition demonstrates our commitment to the expansion of our thrombectomy platform to remove thrombus in venous vascular beds, with an exciting technology that offers significant improvements over current therapies,” said Gary Maharaj, president and chief executive officer of Surmodics.
“The ReVene thrombectomy catheter has the potential to significantly expand the use and accessibility of venous mechanical thrombectomy by allowing physicians to intervene early and complete the procedure in a single session,” said Stephen Black (Guy’s and St Thomas’ NHS Foundation Trust, London, UK), principal investigator and leading enroller of the VETEX feasibility study. “The ease of use, intuitive design, and efficient performance of this device enables it to become the first-line treatment and a confident choice by venous interventionalists.”
Under the terms of the acquisition agreement, Surmodics acquired Vetex with an upfront payment of US$39.9 million. Additional payments of up to US$7 million, US$3.5 million of which are guaranteed, may be made upon achievement of certain product development and regulatory milestones.
Surmodics expects to initiate clinical evaluation activities for the Pounce arterial thrombus retrieval system for removing clot in peripheral arteries in the second half of fiscal 2021 and for the ReVene thrombectomy catheter for removal of clot from veins in fiscal 2022. A projected timeline for further commercialisation will be announced later this fiscal year.












