LimFlow SA today announced publication of positive two-year data from the ALPS registry of the LimFlow percutaneous deep vein arterialisation (pDVA) system. Results were published online yesterday in the Journal of Endovascular Therapy and will also appear in the August print issue of the journal.
The ALPS Registry is the largest study to date of no-option chronic limb-threatening ischaemia (CLTI) patients treated with the LimFlow system reporting mid- and long-term results.
“In this complex group of patients, pDVA using the LimFlow device has shown to be feasible and safe with high technical success and good amputation-free survival at six months up to 24 months coupled with good wound healing,” said the paper’s lead author, Andrej Schmidt, head of the Angiology CathLab at Leipzig University Hospital, Leipzig, Germany. “In selected patients with no-option CLTI, pDVA is a safe and effective treatment to prevent amputation and heal wounds.”
“Patients with this disease suffer with chronic ulcers, often including gangrene, recurrent pain, and a very poor quality of life. CLTI is associated with poor mid- and long-term survival, and no-option patients often face the worst outcomes,” said Roberto Ferraresi, chief of the Peripheral Interventional Unit at the San Carlo Clinic, Milan, Italy, co-author of the paper, and a pioneer in the interventional treatment of CLTI. “These patients desperately need an option to avoid amputation and improve their life, and LimFlow offers a very promising alternative for them.”
The ALPS Registry is a multicentre, retrospective study conducted at centres in Alkmaar, the Netherlands; Leipzig, Germany; Paris, France; and Singapore encompassing 32 end-stage—or “no-option”—CLTI patients.
All patients had non-healing wounds on the target foot and no arterial target for surgical or endovascular revascularisation. Among them, 66% had diabetes, 53% had renal insufficiency, and 16% were dialysis dependent. The majority of patients had experienced failed prior attempts at revascularisation.
The registry reported major amputation-free survival of 84%, 71%, and 67%, respectively, at six months, one year and two years. Additionally, a positive trend of successful wound healing was demonstrated, with 73% of wounds completely healed at two years. Technical success was 97%.
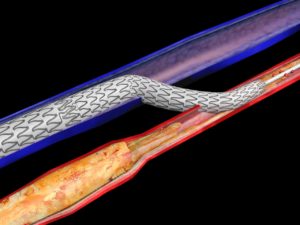
When all other therapeutic options have been exhausted and a CLTI patient is facing major amputation, the minimally-invasive LimFlow system is designed to bypass blocked arteries in the leg and deliver oxygenated blood back into the foot via the veins. For many patients, restoring perfusion in the lower limbs resolves chronic pain, improves quality of life, enables wound healing and prevents major amputation.
“It is very gratifying to see these durable results being demonstrated by the LimFlow system in a real-world setting with consecutive patients,” said LimFlow CEO Dan Rose. “For these no-option patients, the next intervention for their leg was going to be major amputation, with the associated morbidity and high mortality risk that comes with amputation. Instead, two years later, the vast majority of patients are alive, with almost three-quarters of them experiencing completely healed wounds. This registry adds to the building clinical evidence that LimFlow therapy is an effective solution for no-option patients.”
Click here to view a video of how the LimFlow System works.












